-
ISM5411是一款口服PHD特异性抑制剂,可通过诱导肠道屏障保护基因的表达来促进炎症性肠病的治疗;
-
ISM5411也是英矽智能自主研发的AI管线中,第五个进入临床阶段的新药项目,目前已在澳大利亚完成首批健康受试者给药;
-
为了在更广大的群体中对ISM5411进行评估,英矽智能计划在1a期临床试验完成后,招募溃疡性结肠炎患者开展全球多中心1b期临床试验。
中国香港,11月30日,由生成式人工智能(AI)驱动的临床阶段生物科技公司英矽智能宣布,已启动潜在“全球首创”PHD抑制剂ISM5411的临床试验用于炎症性肠病治疗(IBD)。
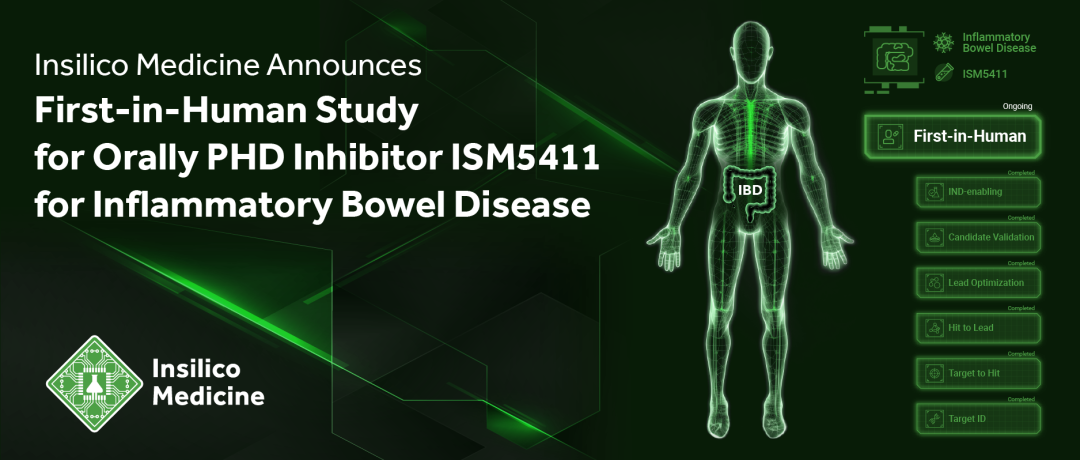
ISM5411由英矽智能自有Pharma.AI药物研发平台赋能设计与开发,是第五个进入临床阶段的AI新药项目。正在进行的1期临床试验(NCT06012578),计划招募76名健康受试者,旨在评估口服候选药物在递增剂量下的安全性、耐受性、药代动力学特征与食物效应,现已在澳大利亚完成首批健康受试者给药。为了在更广大的群体中对ISM5411进行评估,英矽智能计划在1a期临床试验完成后,招募溃疡性结肠炎患者开展全球多中心1b期临床试验。
英矽智能首席医学官Sujata Rao博士负责牵头该项临床试验内部进展,她表示,“炎症性肠炎困扰全球大量患者,而治疗方案相对有限。我们相信,处在临床阶段的这款潜在‘全球首创’疗法有望带来新的选择。”
IBD是一种慢性胃肠道炎症性疾病,影响着全球大量人口,仅在美国就有200万成年患者。IBD包括溃疡性结肠炎(UC)和克罗恩病(CD),这两种类型目前都无法治愈且会使罹患结直肠癌风险的提升达四倍。当前,IBD的标准治疗主要是使用抗炎药物,但这些药物对粘膜修复的改善有限。然而,遗传和临床证据显示,粘膜修复与良好预后密切相关。
研究表明,PHD2在调节缺氧诱导引子1α(HIF-1α)的稳定性和转录活性方面起着关键作用,而HIF-1α可驱动肠道屏障保护基因表达。此外,PHD 在 IBD 患者中明显上调,并与炎症细胞因子和细胞凋亡标志物的表达密切相关。这均表明PHD是治疗IBD 的潜力靶点。
2022年1月,英矽智能宣布提名靶向PHD的ISM5411作为临床前候选化合物,用于IBD治疗。该候选药物由英矽智能自有分子生成引擎Chemistry42辅助开发,是一款肠道限制性口服小分子抑制剂,具有新颖分子骨架和独特结合模式。在临床前研究中,ISM5411作为一种口服肠道限制性小分子抑制剂,表现出良好的安全性和显著的抗结肠炎疗效。
英矽智能联合首席执行官兼首席科学官任峰博士表示,“不断上升的IBD发病率是全球医疗系统的沉重负担,而以人工智能为驱动从头设计的ISM5411以独特的屏障保护增强机制靶向粘膜修复,具有高度创新性。我们将尽力高效推进ISM5411临床试验,尽快满足未竟临床需求。”
以生成式工智能为驱动,英矽智能持续探索人工智能与前沿技术领域交叉。在链接生物学、化学、临床开发的自有Pharma.AI药物研发平台驱动下,英矽智能现已搭建涵盖30余个创新项目的多元化AI药物管线,其中5个项目已经到达临床阶段。近期,英矽智能启动位于加拿大蒙特利尔的AI研发中心,进一步强化内部AI算发研发与平台搭建能力。
英矽智能创始人兼首席执行官Alex Zhavoronkov博士表示,“在英矽智能,我们期待将人工智能技术和人类智能创新有机结合造福全球病患。此次进入临床阶段的IBD项目再次验证了英矽智能运用AI算法赋能难治疾病疗法研究与开发的能力。”
关于英矽智能
英矽智能是一家由生成式人工智能驱动的临床阶段生物医药科技公司,通过下一代人工智能系统连接生物学、化学和临床试验分析,利用深度生成模型、强化学习、转换模型等现代机器学习技术,构建强大且高效的人工智能药物研发平台,识别全新靶点并生成具有特定属性分子结构的候选药物。英矽智能聚焦癌症、纤维化、免疫、中枢神经系统疾病、衰老相关疾病等未被满足医疗需求领域,推进并加速创新药物研发。
更多信息,请访问网站
www.insilico.com
商务合作,请联系 bd@insilico.ai
媒体垂询,请联系 pr@insilico.ai
右滑查看中文版
-
ISM5411 is an oral, PHD-specific inhibitor that facilitates the treatment of IBD by inducing the expression of gut barrier protective genes.
-
ISM5411 is Insilico Medicine’s fifth AI-driven drug program to enter clinical trials, with the first dose of healthy subjects completed in Australia.
-
To further evaluate ISM5411 in wider populations, Insilico plans to conduct global, multi-center Phase Ib trials in patients with ulcerative colitis following the Phase Ia study.
New York and Hong Kong – Insilico Medicine (“Insilico”), a generative artificial intelligence (AI)-driven clinical-stage biotechnology company, today announced that the company has initiated the first-in-human study for ISM5411, a potentially first-in-class PHD inhibitor for the treatment of inflammatory bowel disease (IBD).
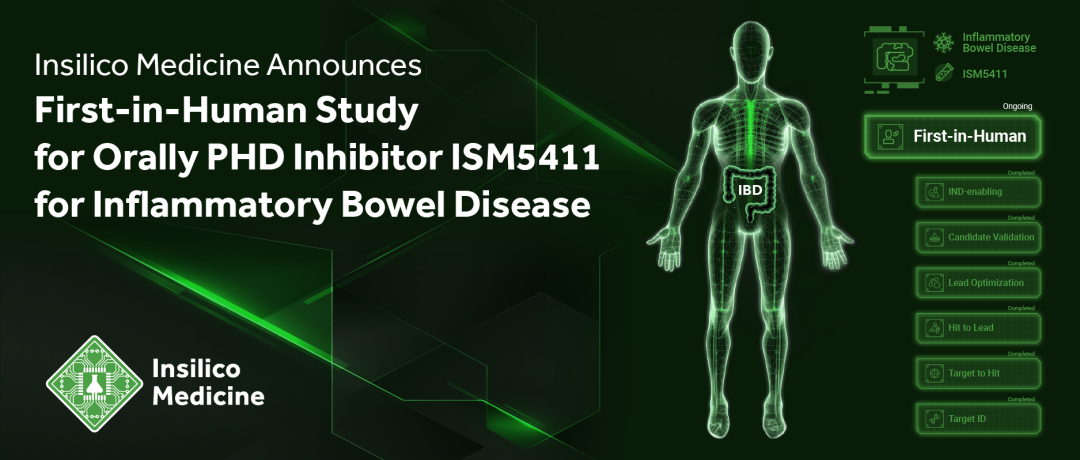
Developed by Insilico’s proprietary end-to-end AI drug discovery platform, Pharma.AI, ISM5411 is Insilico’s fifth AI drug program to enter the clinic. The ongoing Phase I study is designed to evaluate the safety, tolerability, pharmacokinetics and food effects of ISM5411 at increasing oral doses in 76 healthy subjects, with the first dose in healthy volunteers now completed in Australia. To further evaluate ISM5411 in wider populations, Insilico plans to conduct global multi-center Phase Ib trials in patients with ulcerative colitis following the Phase Ia study.
“IBD impacts a large patient population who currently have limited therapeutic options,” says Sujata Rao, MD, Chief Medical Officer at Insilico Medicine who is leading the clinical trial. “We believe this new treatment – which is potentially first-in-class – could offer a promising alternative.”
IBD is a chronic inflammatory disease of the gastrointestinal tract, including ulcerative colitis (UC) and Crohn’s disease (CD), that affects huge populations globally, and 2 million adults in the U.S. alone. Neither disorder is curable, and they both increase the risk of colorectal cancer up to four times. The current standard of treatment for IBD consists of anti-inflammatory drugs which offer limited improvement in mucosal healing, which is closely correlated to positive prognosis according to genetic and clinical evidence.
Research shows that PHD plays a critical role in regulating the stability and transcriptional activity of the hypoxia-inducible factor-1α (HIF-1α), which drives the expression of barrier protective genes. PHD is significantly up regulated in IBD patients, and shows strong correlation with the expression of inflammatory cytokines and the apoptosis marker, suggesting that PHD is a potential target for IBD treatment.
In January 2022, Insilico announced the nomination of ISM5411 as a preclinical drug candidate (PCC) for the treatment of IBD targeting PHD. ISM5411 was developed by Chemistry42, Insilico’s generative AI engine for molecule development, and features a novel scaffold and a unique binding mode. In preclinical studies, ISM5411 demonstrated a favorable safety profile and significant anti-colitis efficacy as an oral, gut-restricted small molecule inhibitor.
“The rising incidence of IBD places a heavy burden on healthcare systems globally,” said Feng Ren, PhD, co-CEO and Chief Scientific Officer of Insilico Medicine. “Designed by AI from scratch, ISM5411 targets mucosal healing with the unique mechanism of barrier protection enhancement, which could potentially lead to innovative clinical options for IBD patients. We are committed to accelerating ISM5411 clinical trials to reach unmet clinical needs as soon as possible.”
Driven by generative artificial intelligence, Insilico stands at the intersection of artificial intelligence and state-of-art technologies. The company has developed a comprehensive portfolio of more than 30 pipelines with 5 programs currently in the clinical stage developed using Pharma.AI, an integrated AI-driven drug discovery platform connecting biology, chemistry, and clinical development. Recently, Insilico launched the Company’s Al R&D Center in Montreal, Canada to further strengthen its AI algorithm research and platform building capabilities.
“At Insilico, we are driven to help patients in need using state-of-the-art AI technology combined with human ingenuity,” said Alex Zhavoronkov, PhD, Founder and co-CEO of Insilico Medicine. “This IBD program is the latest example of how we are using our AI algorithms to advance new cures for difficult-to-treat diseases.”
About Insilico Medicine
Insilico Medicine, a global clinical stage biotechnology company powered by generative AI, is connecting biology, chemistry, and clinical trials analysis using next-generation AI systems. The company has developed AI platforms that utilize deep generative models, reinforcement learning, transformers, and other modern machine learning techniques for novel target discovery and the generation of novel molecular structures with desired properties. Insilico Medicine is developing breakthrough solutions to discover and develop innovative drugs for cancer, fibrosis, immunity, central nervous system diseases, infectious diseases, autoimmune diseases, and aging-related diseases. www.insilico.com



 ufabet
มีเกมให้เลือกเล่นมากมาย: เกมเดิมพันหลากหลาย ครบทุกค่ายดัง
ufabet
มีเกมให้เลือกเล่นมากมาย: เกมเดิมพันหลากหลาย ครบทุกค่ายดัง


